RBQM Essentials – Black Belt, Instructor-led
Elevate Your Expertise: Discover the Power of RBQM Essentials Black Belt Training
“We are happy with the engagement and level of knowledge that MyRBQM Academy brought to our Operations teams as to the RQBM implementation. And, by using Cyntegrity’s unique RBQM technology we were able to retrospectively analyze a real world clinical study and conduct a workshop to successfully generate new ideas for further improvements on a global scale,” said Jens Opitz, Executive Director, Head of Global Clinical Operations at Merz Pharmaceuticals GmbH. Read also: Merz Pharma’s RBQM Journey published by Applied Clinical Trials
Designed for advanced clinical research professionals, this course will empower you to lead clinical trials and drive organizational change with confidence and expertise.

Overview
Welcome to RBQM Essentials’ Black Belt’: Master Advanced RBQM Strategies & Change Management
Perfect your clinical research skills with RBQM Essentials Black Belt training! It is designed for professionals seeking expertise in advanced RBQM strategies and change management. This comprehensive course, customized for various clinical research roles, includes 4 interactive two-hour modules that explore designing KRIs & QTLs, central monitoring, RBQM facilitation in clinical trials, and change management.
Dynamic Learning: Master Advanced RBQM and Change Management
Our engaging instructor-led workshops mix advanced lessons, case studies, and hands-on sessions, allowing you to apply RBQM and change management in your daily practice. Benefit from fellow participants’ diverse experiences and expert trainers as you collaborate and share knowledge in real time.
Expert Guidance and Knowledge Sharing
Our experienced industry professionals enrich workshops by sharing invaluable real-life experiences, including live demonstrations of Risk-Based Quality Management enabling technologies. Embrace this unique learning opportunity and elevate your RBQM expertise with Black Belt training!
Certification and Mastery: Enhance Your Professional Credentials
After completing the Black Belt course and passing a required quiz, you’ll earn your Black Belt certification. It showcases your expertise in advanced RBQM strategies and change management. For those pursuing Black Belt Mastery, successfully completing an optional risk-assessment assignment will further demonstrate your proficiency and commitment to the field.
Join MyRBQM Academy Alumni Community
Becoming a certified Black Belt, you’ll join an exclusive community of MyRBQM Academy alumni, growing your professional network and fostering collaboration with like-minded individuals. Utilize this supportive network to further your career and contribute to a thriving clinical research landscape.
Transform Your Clinical Research Career
RBQM Essentials Black Belt training equips you with the knowledge and skills to lead teams, analyze risk signals, and drive change effectively in your organization. Excel in your clinical research role and play a pivotal part in clinical trials’ successful execution. Enroll in the Black Belt course today and make a lasting impact on your career and the clinical research industry.
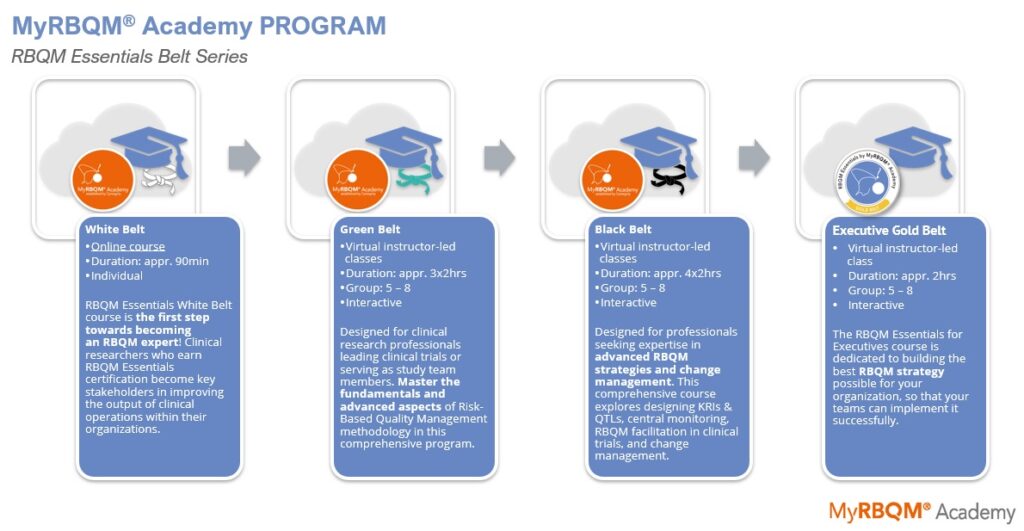
BB Module 1: Designing KRIs & QTLs; Metrics, Thresholds, and Signals
- Instructor-led training of four 2-hour online sessions (M1-4)
- General Overview
- Designing KRIs
- Thresholds
- QTLs
BB Module 2: Central Monitoring During a Study
- KRIs
- QTLs
- Central Statistical Monitoring
- Handling (Potential) Issues
- Influencing Others
BB Module 3: Facilitating RBQM in Clinical Trials
- The Role of Facilitating
- Risk Assessment
- Risk Control
- Don’t Forget!
BB Module 4: Change Management
- The (in)Famous Change Curve
- The Adaptable Human
- Change versus Transition
- Change Management
- Phases of Change and Common Behaviors
- Dealing with Resistance
- RBQM Implementation: Take a Phased Approach
Optional: Black Belt Mastery Certification Project
Who should attend?
- Risk Managers
- Roles implementing RBQM at a company
- Study Managers
- Central Monitors
- Lead Biostatisticians
- Lead Data Managers
- Medical Monitors
- Roles responsible for programming KRIs/QTLs
(Note: Prior MyRBQM Academy White Belt and Green Belt certification are required; however, these prerequisites can be skipped if you successfully pass the prequalification quiz, allowing you to enroll in the Black Belt course directly.)
Why should you attend?
- Elevate Your Expertise: Become a certified Black Belt and accelerate your career in clinical research, showcasing your advanced knowledge in RBQM and change management.
- Ensure Compliance: Master the techniques to achieve regulatory compliance in your industry, protecting your organization’s reputation and integrity.
- Exclusive Access: Join the prestigious MyRBQM Academy Alumni community, connecting with industry peers and thought leaders who are equally committed to excellence.
- Bonus Material: Enhance your learning journey with a valuable bonus – receive the Adaptive Monitoring for Clinical Trials (Flowcharts) free of charge, giving you a comprehensive overview of quality risk management in a visually engaging format.
- Comprehensive Curriculum: Immerse yourself in 3 interactive sessions of 2 hours each, covering essential topics such as KRIs & QTLs, central monitoring, facilitating RBQM in clinical trials, and change management.
- Unleash Your Potential: By attending this Black Belt training, you’ll be empowered to make a meaningful impact in your organization, driving improvements in overall quality and profitability.
Pricing
For You
€875,00 – €6.475,00
- 2 year access
- 100% online
- Unique additional tools






