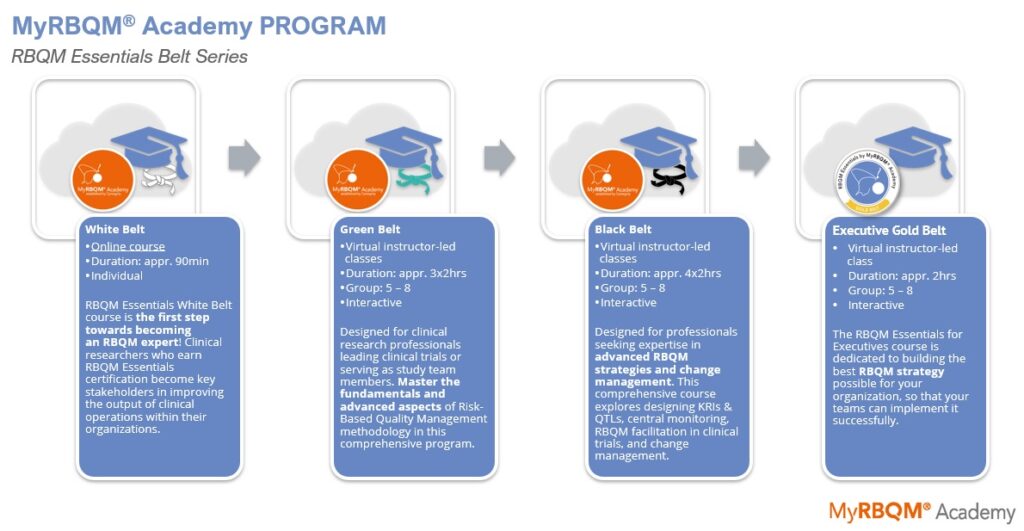
RBQM Essentials – White Belt, e-Course
Get to grips with the fundamentals of RBQM with this self-paced course, the first of the RBQM Essentials Series.

Overview
The RBQM Essentials White Belt course is dedicated to the fundamental RBQM knowledge every risk-oriented clinical expert must have.
Enroll in RBQM Essentials White Belt course and take a first step towards becoming an RBQM expert! Clinical researchers who earn RBQM Essentials certification become key stakeholders in improving the output of clinical operations within their organizations. They strive to optimize data quality and patient safety in clinical trial conduct by implementing risk-based processes and establishing metrics like KRIs, KPIs, and QTLs, that minimize the potential for error, misconduct and fraud.
RBQM White Belt course is the perfect starting point for Clinical Research Professionals to gain a solid foundation in Risk-Based Quality Management. This course is the first of three levels in our RBQM Essentials series, which also includes the Green Belt and Black Belt courses. Attendees include Clinical Project Managers, Clinical Operations Managers, Clinical Data & Quality Managers, CRAs & Monitors, and all other Clinical Research Professionals.
This course offers attendees the opportunity to earn a career certificate and advance their career in the clinical research industry. Additionally, it provides essential compliance training to ensure that professionals understand and adhere to RBQM regulations within their organization.
As an RBQM White Belt course attendee, you will also join our exclusive MyRBQM Academy Alumni community! It provides access to unique bonus materials and further networking opportunities.
Throughout the course, attendees will cover a variety of topics, including understanding RBQM, the purpose and importance of RBQM, and the key aspects of an RBQM. Additionally, participants will learn about the different clinical risk categories and types, as well as the methods, roles, and types of monitoring, including Targeted and Triggered Monitoring. The course also covers Quality by Design, and the development and implementation of risk-based quality management.
By attending our RBQM White Belt course, you will gain essential knowledge and skills needed to take your clinical research career to the next level. Sign up today and join the ranks of RBQM professionals.
Lecture 1: Understanding RBQM
- Introduction to RBQM Essentials White Belt course
- What is Risk-Based Quality Management?
- Why is RBQM essential to successful clinical trial conduct?
- What is the main purpose of RBQM?
Lecture 2: Taking a ‘Risk-Based Approach’
- What does it mean ‘taking a risk-based approach’?
- What are the new risk-based Monitoring methods, types and roles?
- What is the difference between Targeted and Triggered Monitoring?
- What are the key aspects of an RBQM Plan?
Lecture 3: Perspectives to Approach Risk
- What is ‘Quality by Design’?
- What is the concept of ‘Design Space’?
- What is ‘risk’ and its properties?
- What are the different clinical risk categories and types?
Lecture 4: Practical Considerations
- What does it take to develop and implement risk-based quality management?
- Who are involved in RBQM?
- How can workflows be managed?
- What is ‘holistic RBQM’?
Lecture 5: The RBQM Toolbox
- What are the key elements of RBQM implementation?
- Which tools and technologies are ‘fit-for-purpose’?
- What are the benefits of specialized RBQM tools?
Lecture 6: Let’s Ask the Experts
- RBQM Myth 1 – 5 (popular misconceptions)
Who should attend?
- Clinical Project Managers
- Clinical Operations Managers
- Clinical Data & Data Quality Managers
- CRAs & Monitors
- … and all other Clinical Research Professionals
Why should you attend?
- Earn a career certificate to advance your career
- Achieve compliance in your organization and industry
- Join the exclusive MyRBQM Academy Alumni community
- Access to unique bonus material
Pricing
For You
€190,00
- 2 year access
- 100% online
- Unique additional tools
*Courses are limited to 1 item per person. For purchasing for a team, please get in touch.




