RBQM Protocol Design: Why Monitoring Starts Before First Patient

Risk-based quality management begins in protocol design. See how ICH E6(R3) shifts oversight from monitoring plans to decision logic.
Decentralized Clinical Trials Outlook in the USA
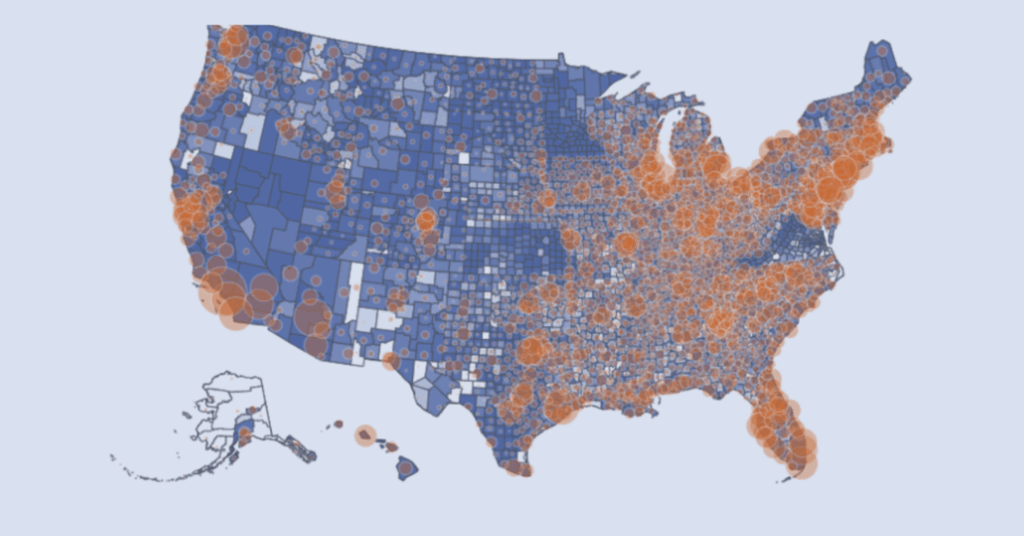
Interactive visualizer exploring patient distribution, oncology expertise, and broadband access to support understanding of decentralized trial feasibility in the USA.
Site Readiness Under ICH E6(R3)

Key expectations for site readiness under ICH E6(R3), including documentation, risk-based oversight, and digital data governance.
ICH E6(R3) Final Guideline: What You Need to Know

ICH E6(R3) is now adopted. Learn what changes for sponsors, CROs, and sites, including accountability, CtQ planning, proactive monitoring, and participant protection.
Site Readiness Tips

Practical steps trial sites can take to align with ICH E6(R3) expectations for training, data governance, and risk-based monitoring.
Sponsor Responsibilities in ICH E6(R3)

What E6(R3) means for sponsor accountability, oversight, and documentation — plus a practical sponsor responsibilities infographic.
ICH E6(R3) vs ICH E8(R1): How They Work Together

How ICH E6(R3) and E8(R1) work together to support quality-by-design, risk-based oversight, and improved patient protection in clinical trials.
R&D Efficiency: Big Pharma Overview
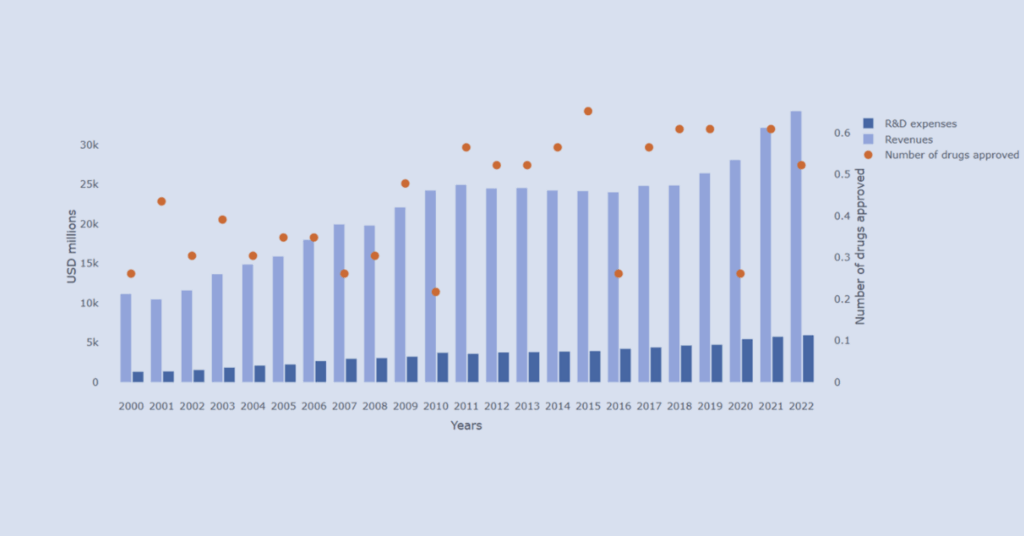
Explore R&D investment and innovation output across leading pharmaceutical companies through an interactive data visualizer.
Economy and Health Indicators Across OECD Countries
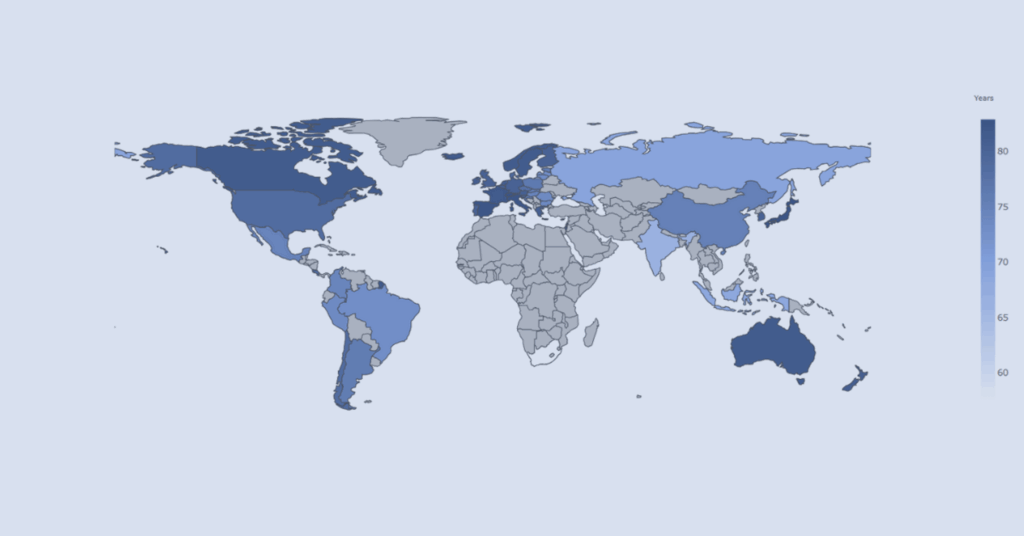
Interactive visualizer comparing economic indicators and health outcomes across OECD countries to support contextual understanding of population health trends.
COVID-19 Impact on Global Clinical Trials
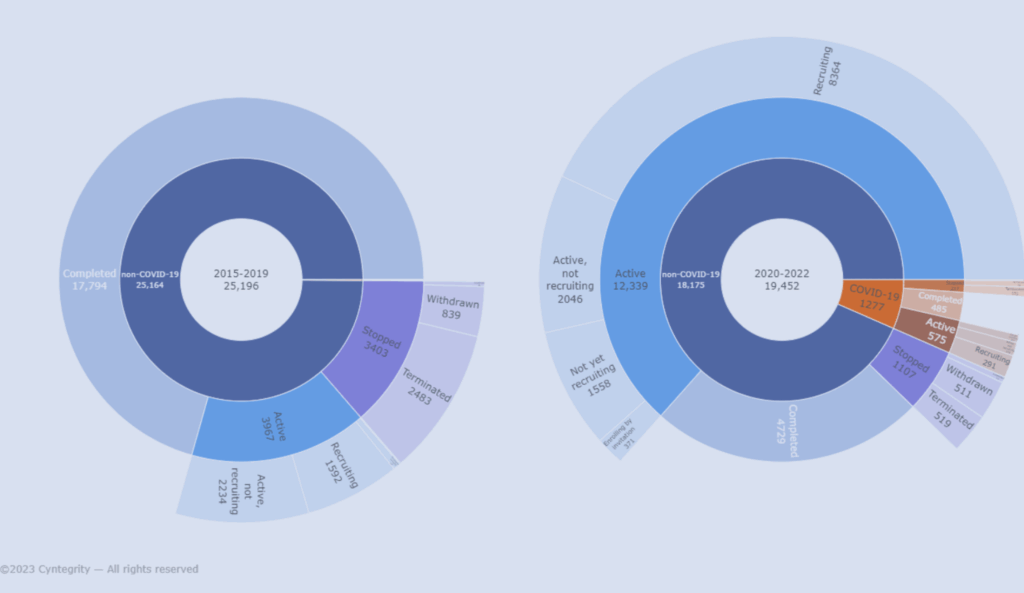
Interactive visualizer showing how COVID-19 affected global clinical trial activity, comparing COVID-19 and non-COVID-19 studies from 2015 to 2022.



