AI Literacy in Clinical Trials Under ICH E6(R3)

AI-supported workflows in clinical trials increasingly fall within GCP, computerized system, data integrity, and AI governance expectations. Sponsors and CROs need teams that understand oversight, validation, accountability, and responsible AI use.
FDA Warning Letter Highlights AI Oversight Risks in Clinical Trials

FDA’s warning letter signals a shift: AI use without oversight is a compliance risk. What clinical trial teams must understand to stay inspection-ready.
Global Uncertainty and Clinical Research: Why a Risk-Based Perspective Matters

Global risks in clinical trials are increasing as geopolitical instability, supply chain disruptions, and regulatory changes affect research worldwide. Learn why RBQM is essential for proactive oversight.
RBQM Protocol Design: Why Monitoring Starts Before First Patient

Risk-based quality management begins in protocol design. See how ICH E6(R3) shifts oversight from monitoring plans to decision logic.
Risk-Based Quality Management in Medical Devices: ISO 14155 vs ICH E6(R3)
Medical device investigations are now assessed like RBQM trials. Learn how ISO 14155 and ICH E6(R3) align and download the practical translation guide.
From EMA Findings to ICH E6(R3): QbD and RBQM in Practice

How EMA inspection findings translate into ICH E6(R3) expectations, and why Quality by Design and RBQM must operate together in clinical trials.
Risk-based quality management in 2026

Risk-based quality management in 2026: how ICH E6(R3) defines RBQM, where practice diverges, and what regulators now expect in clinical trials.
FDA AI Guidance 2025 Interactive Visualizer
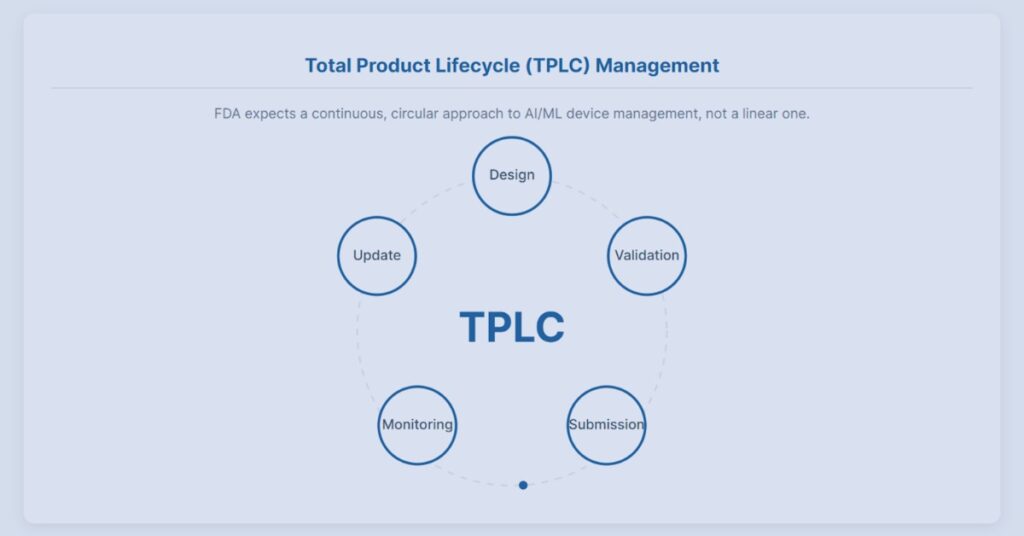
Explore how FDA’s 2025 AI guidance shapes lifecycle oversight for AI-enabled medical software through interactive regulatory scenarios.
ICH E6(R3) Interactive Visualizer: GCP Explained Visually
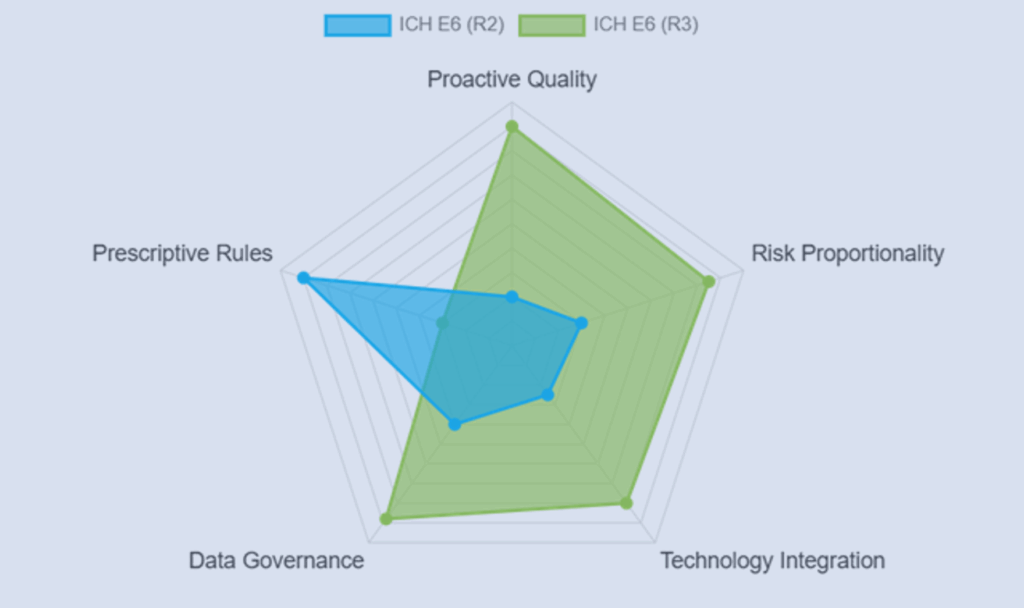
Explore the evolution of Good Clinical Practice with an interactive ICH E6(R3) visualizer showing ethics, risk-based oversight, and modern GCP design.
EMA Clinical Trials Map Visualizer Explained
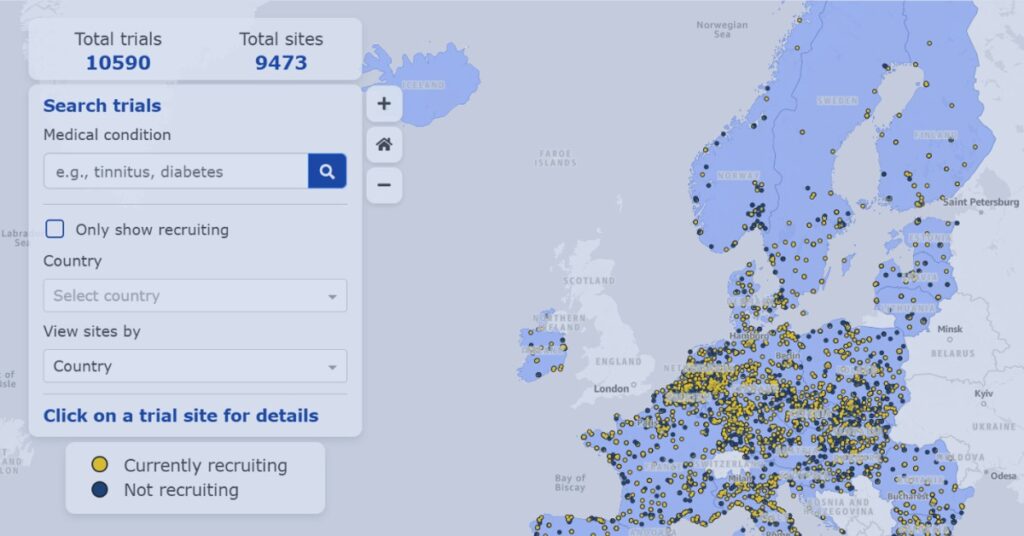
Explore how the EMA Clinical Trials Map visualizes trial activity across Europe to support recruitment planning and site strategy decisions.



