Decentralized Clinical Trials Outlook in the USA
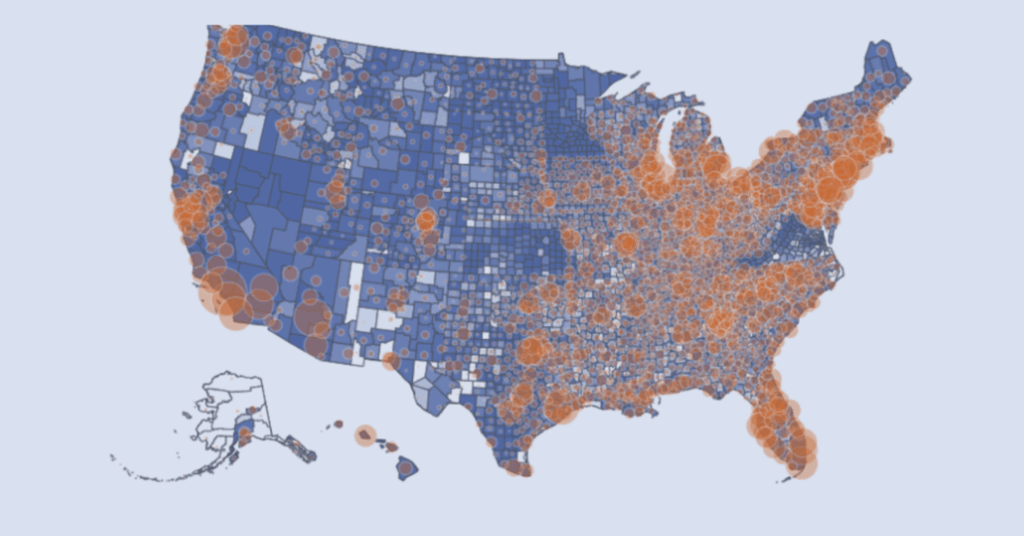
Interactive visualizer exploring patient distribution, oncology expertise, and broadband access to support understanding of decentralized trial feasibility in the USA.
AI-Driven Predictive Analytics in Risk-Based Monitoring

Discover how AI-driven predictive analytics is reshaping risk-based monitoring under ICH E6(R3), enabling earlier intervention and stronger oversight.
The Cost of Poor Quality in Clinical Trials & How to Avoid It

Protocol changes and preventable risks can delay trials and drive major costs. Learn why proactive quality planning and RBQM lead to more predictable outcomes.
Phase III Oncology: Targeted Risk-Based QA Case Study

How ADAMAS used AI-augmented RBQM analytics to strengthen oversight, focus QA effort, and improve inspection readiness in a global Phase III oncology study.
Site Readiness Under ICH E6(R3)

Key expectations for site readiness under ICH E6(R3), including documentation, risk-based oversight, and digital data governance.
ICH E6(R3) Final Guideline: What You Need to Know

ICH E6(R3) is now adopted. Learn what changes for sponsors, CROs, and sites, including accountability, CtQ planning, proactive monitoring, and participant protection.
Site Readiness Tips

Practical steps trial sites can take to align with ICH E6(R3) expectations for training, data governance, and risk-based monitoring.
Sponsor Responsibilities in ICH E6(R3)

What E6(R3) means for sponsor accountability, oversight, and documentation — plus a practical sponsor responsibilities infographic.
Milestone | We Have Just Received our ISO 9001:2015 Certificate

Cyntegrity is now ISO 9001:2015 certified. Learn how this strengthens quality governance, continuous improvement, and trust in AI-augmented RBQM solutions.
ICH E6(R3) vs ICH E8(R1): How They Work Together

How ICH E6(R3) and E8(R1) work together to support quality-by-design, risk-based oversight, and improved patient protection in clinical trials.



