Deliver sponsor-ready oversight, prove quality, and reduce operational costs, all in one platform
Deliver sponsor-ready oversight, prove quality, and reduce operational costs, all in one platform.
35%
Fewer inspection-prep hours
2×
Faster inspection-plan creation
≤4h
Data routed to dashboards
Centralized oversight and predictive alerts help you cut costs, boost quality, and prove it to sponsors.
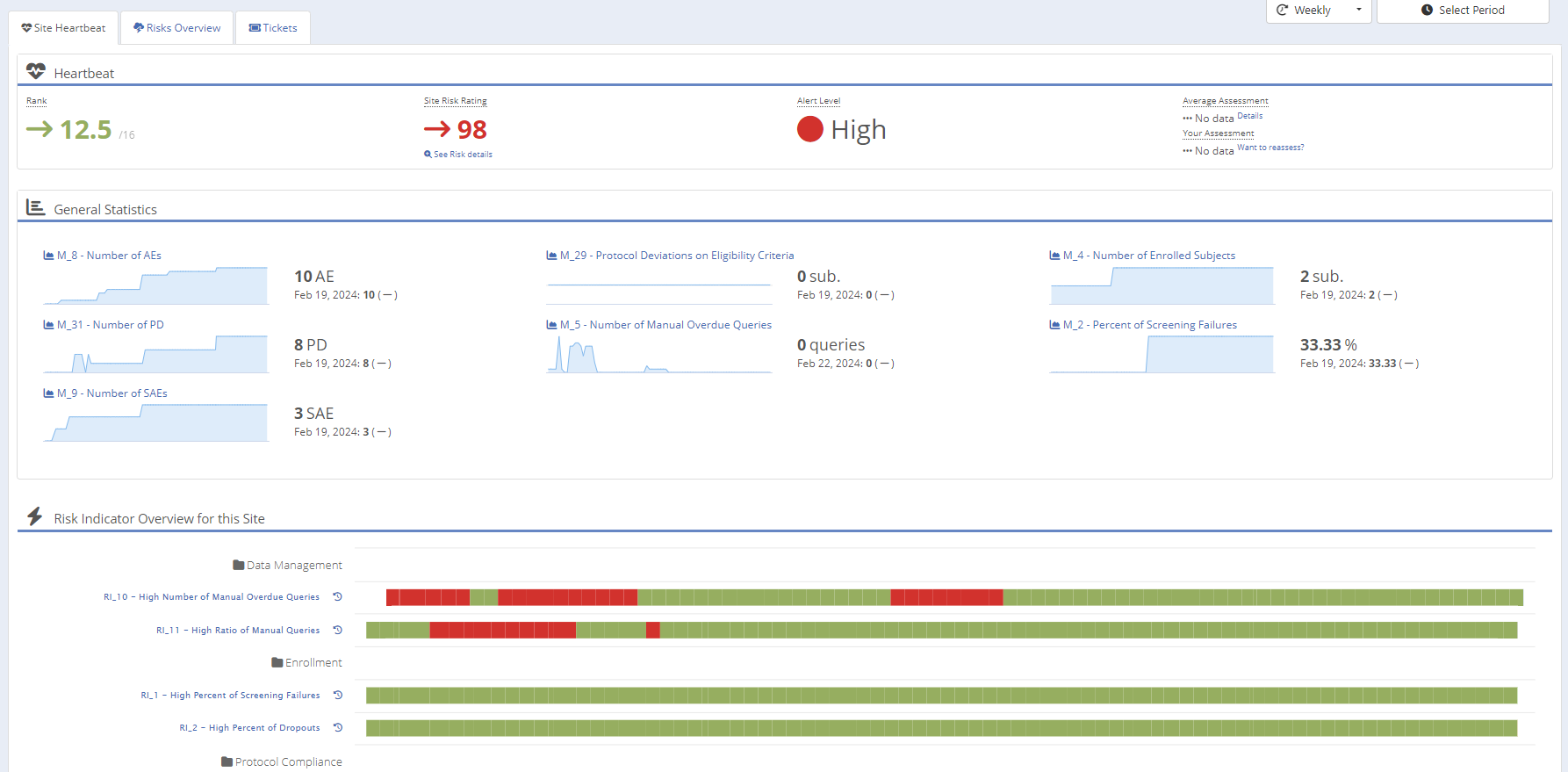
Instant visibility into site performance, combining signals across risk indicators, deviations, and queries — ready for sponsors at any time.
Weighted risk ratings and alert levels identify issues earlier and support faster, defensible escalation decisions.
Live counts and trend graphs for AEs, PDs, SAEs, enrollment, queries — always updated, always audit-ready.
Visual trend detection across key categories — showing sponsors exactly how your team identifies signals earlier.
Uniform categories and risk criteria ensure repeatable RBQM delivery across all studies, reducing variability and audit findings.
Drill into trend behavior instantly — enabling faster root-cause analysis and supporting audit defensibility.

Trusted by CROs who must deliver consistent, sponsor-ready RBQM quality at scale, with standardized templates, predictive alerts, and transparent oversight.




Do you need to prove oversight excellence to win sponsor contracts?
Is inspection-readiness a priority for every project?
Are you under pressure to reduce monitoring costs without sacrificing quality?
Design out avoidable risk before FPFV. AI-augmented protocol analysis surfaces CtQs, quantifies complexity, predicts deviation hot-spots, and reduces amendment drag, so you lock in patient-safe, inspection-ready design from day one.
Operationalize RBQM with verifiable math. AI-guided RACTs, calculation transparency, RBAC-secured workflows, and cross-study analytics deliver proportionate controls, clear KRI/QTL governance, and faster, defensible decisions at scale.
Move beyond manual cleaning and fragmented reviews. With time-series anomaly detection, protocol-aware deviation checks, and structured reasoning, the MyRBQM® Portal highlights what truly requires attention.
Stay ahead of issues, not behind them. Portfolio-wide, AI-enhanced dashboards fuse KRIs, QTLs, and deviation signals to cut escalation latency and focus monitors on high-value actions, fully aligned with ICH expectations.
See the full patient story—fast. Subject Profiles synthesize visits, AEs, ConMeds, labs, and trendlines, accelerating case review and improving safety decisions with transparent traceability and RBAC-controlled access.
Fast, secure connectivity with your clinical platforms through ready-made endpoints and low-overhead, real-time or batch data sync.
Pre-engineered, validated connectors to major EDC systems — reducing integration time while enabling secure, real-time or scheduled data sync.
Seamlessly connect your enterprise data platforms with secure, governed, low-overhead sync — without restructuring your existing data lake.
Our platform and processes meet internationally recognized security, availability, and confidentiality standards. ISO 27001:2022 certification and SOC 2 examinations provide independently verified assurance of our information security controls.
Our clinical-grade AI is independently certified through Microsoft’s Healthcare AI program, ensuring transparent, human-guided oversight, responsible AI practices, and secure integration with Azure’s validated infrastructure.
The MyRBQM Portal is built in alignment with 21 CFR Part 11 and GAMP 5, ensuring data integrity, traceability, and suitability for use in regulated clinical research environments.
Download the guide to delivering sponsor confidence, audit readiness, and operational efficiency with RBQM.

Industry Solutions
Featured Insights

Start Your Roll-Out

RBQM Capability Pathway
Quick Answers
