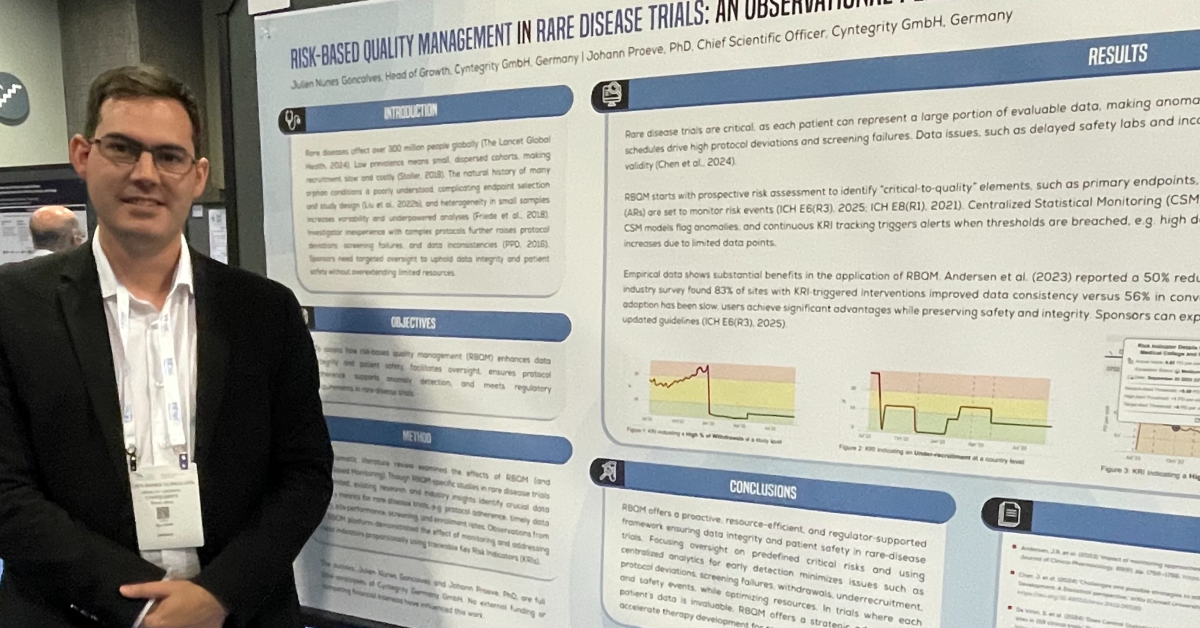
Key Questions to Ensure Your RBQM System Meets Regulatory Standards
When evaluating Risk-Based Quality Management (RBQM) systems, asking the right questions is crucial to ensure you choose a solution that meets your needs and aligns with regulatory guidelines. Here are five key questions to consider, along with how Cyntegrity’s MyRBQM® Portal excels in each area:
Yes, our portal captures datasets with multiple timestamps, allowing you to see exactly how data looked at any point in the past. This transparency ensures you can track the progress of your study based on the actions taken at that time. For example, you can see how a site’s performance has changed over the last week, month, or year.
Yes, we use three timestamps per data point—when the data was recorded, when it was imported, and when it was appended to the record. This ensures that every piece of data is verifiable and can be traced back to its original context. This means you can always understand why specific decisions were made at any given time.
Yes, our system documents all data transformations, making it easy to trace the data flow and understand the changes applied. This ensures the data is fully auditable, allowing you to reproduce any analysis done at a particular point in time. For instance, you can show why a decision was made based on the data available at that moment.
Yes, our platform allows multiple users to work on a clinical study simultaneously. This collaborative functionality ensures teams can efficiently manage and analyze data without delays, enhancing productivity and decision-making. For example, different team members can update and review data in real-time, ensuring everyone is on the same page.
Yes, our portal is designed to handle data efficiently and accurately, even under heavy use. Our robust data processing tasks and efficient study database queries ensure that the system remains reliable and effective, providing decision-makers with the necessary confidence. This means you can trust the system to perform well, even during peak times.
Final Thought
When it comes to choosing an RBQM system, an expert system, like the MyRBQM Portal, should stand out for its compliant design, transparency, verifiability, auditability, and collaborative functionality. These qualities ensure that you have a reliable and comprehensive tool for risk-managing clinical trials, making it the superior choice for decision-makers in the BioPharma market.